AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Taurine effects on body9/23/2023 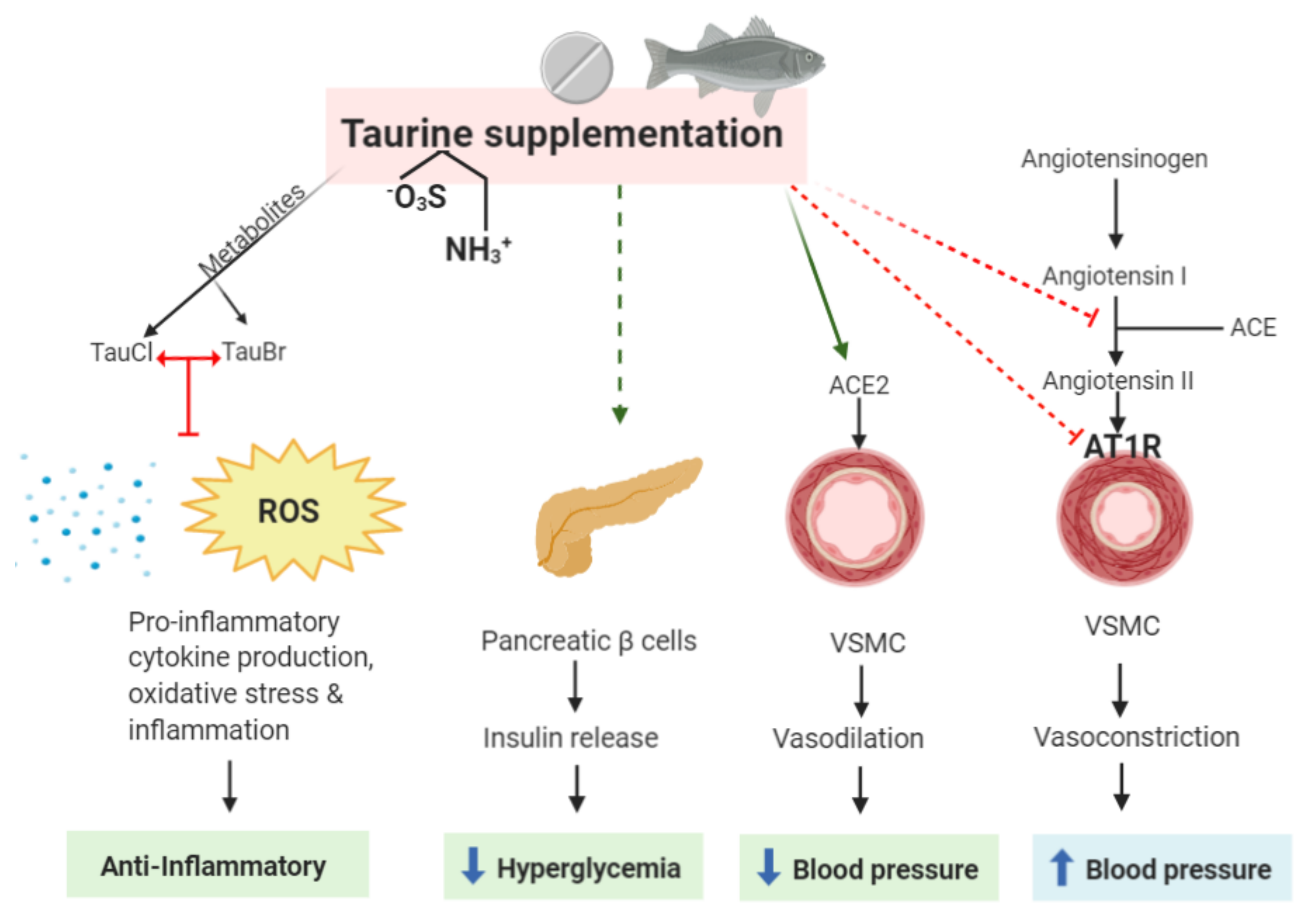 
In 1993, about 5,000–6,000 tonnes of taurine were produced for commercial purposes: 50% for pet food and 50% in pharmaceutical applications. A direct approach involves the reaction of aziridine with sulfurous acid. Synthetic taurine is obtained by the ammonolysis of isethionic acid (2-hydroxyethanesulfonic acid), which in turn is obtained from the reaction of ethylene oxide with aqueous sodium bisulfite. The sulfonic acid has a low p K a ensuring that it is fully ionized to the sulfonate at the pHs found in the intestinal tract. Taurine exists as a zwitterion H 3N +CH 2CH 2SO − 3, as verified by X-ray crystallography. It is an unusual example of a naturally occurring sulfonic acid. Taurine concentrations in land plants are very low or undetectable, but up to 1000 nmol/g wet weight have been found in algae. It is essential for cardiovascular function, and development and function of skeletal muscle, the retina, and the central nervous system. This compound has many biological roles, such as conjugation of bile acids, antioxidation, osmoregulation, membrane stabilization, and modulation of calcium signaling. It was discovered in human bile in 1846 by Edmund Ronalds. Taurine is named after Latin taurus ( cognate to Ancient Greek ταῦρος, taûros) meaning bull or ox, as it was first isolated from ox bile in 1827 by German scientists Friedrich Tiedemann and Leopold Gmelin. It is a major constituent of bile and can be found in the large intestine, and accounts for up to 0.1% of total human body weight. Taurine ( / ˈ t ɔː r iː n/), or 2-aminoethanesulfonic acid, is a non-proteinogenic amino sulfonic acid that is widely distributed in animal tissues.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
